What Is Male Infertility?
The World Health Organization (WHO) classifies male infertility as a disease of the male reproductive system — specifically, the inability to achieve pregnancy in a fertile female partner despite 12 or more months of regular, unprotected sexual intercourse.[^1]
When to Seek Help
Situation | When to See a Specialist |
A couple trying to conceive | After 12 months without success |
Female partner age 35+ | After 6 months of trying |
Known male risk factors | Seek evaluation sooner |
Sexual function difficulties | See a doctor promptly |
Source: WHO Infertility Fact Sheet (2023)[^1] and ASRM Practice Committee (2023).[^4]
This diagnosis carries no reflection on a man’s character or worth. Instead, it represents a medical issue with discoverable origins and, more often than not, successful interventions.
Why should both partners undergo testing?
Male and female factors share roughly equal responsibility for fertility challenges. Evaluating only one partner may overlook up to half of all contributing causes — an oversight that costs valuable time and resources.
→ Learn more: Infertility
What Causes Male Infertility?
Problems with sperm production, delivery, or function all contribute to male infertility. The reassuring reality: in most cases, underlying causes can be identified, enabling targeted treatment approaches.
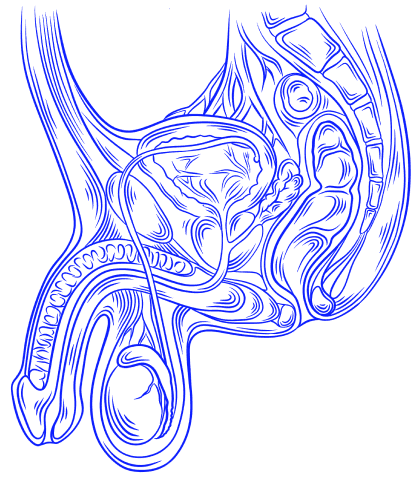
Image: Male Reproductive System — Testes, Epididymis, Vas Deferens, and Prostate
Cause Category | Percentage of Cases |
Testicular factors (problems with sperm production) | 65–80% |
Post-testicular factors (obstruction, ejaculatory problems) | Approximately 5% |
Pre-testicular factors (hormonal imbalances) | 2–5% |
Idiopathic (unknown cause) | 10–20% |
Source: Winters BR, Walsh TJ. The epidemiology of male infertility. Urol Clin North Am. 2014.[^16]
Testicular Causes
The testes harbor the root cause in the majority of male infertility cases. These conditions directly affect the body’s capacity to produce healthy sperm.
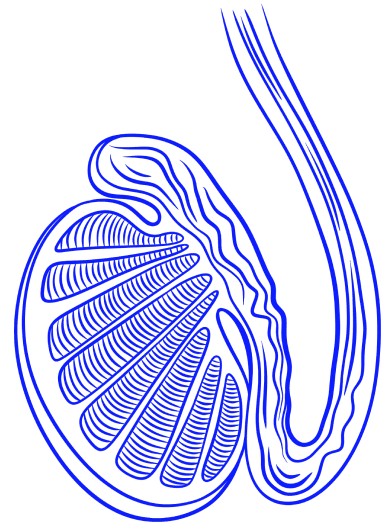
Image: Testis — Internal Structure and Sperm Production
Varicocele
A varicocele occurs when veins draining blood from the testicle become enlarged — think of them as varicose veins within the scrotum. This condition stands as the leading correctable cause of male infertility.
Present in approximately 15% of the general male population, varicoceles appear in up to 40% of men seeking fertility treatment.[^6]
Not all varicoceles affect fertility, but clinically significant varicoceles are associated with abnormal semen parameters — such as lower sperm concentration, reduced motility, decreased normal morphology, and increased DNA fragmentation.
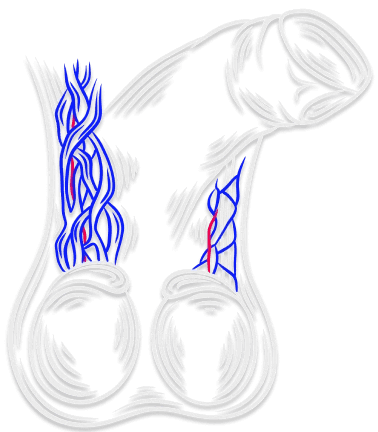
Image: Varicocele — Enlarged Scrotal Veins (Left) vs. Normal (Right)
Genetic Causes
In men with severe sperm production disorders (azoospermia or severe oligozoospermia), approximately 10–15% of cases can be traced to chromosomal abnormalities or gene mutations.[^10]
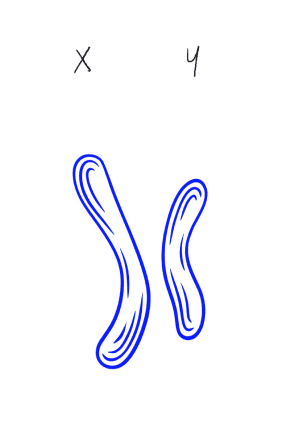

Image: Klinefelter Syndrome (47, XXY) — Normal XY (Left) vs. Abnormal XXY Chromosomal Pattern (Right)
This is significantly higher than in the general fertile male population, where chromosomal abnormalities account for only about 0.6%.[^10]
The primary genetic factors include:
Klinefelter syndrome (47, XXY) — The most common chromosomal cause, occurring in approximately 1 in 600 males. Characteristic features include small, firm testes and often absent sperm in the ejaculate. Modern reproductive techniques may achieve successful sperm retrieval in 40–60% of affected men.[^10]
Y-chromosome microdeletions — Detected in 10–15% of men with non-obstructive azoospermia or severely reduced sperm counts. These deletions affect genes essential for sperm development. Prognosis varies by deletion location.[^4]
CFTR gene mutations — May cause congenital bilateral absence of the vas deferens (CBAVD), leading to obstructive azoospermia. Genetic counseling is recommended.[^4]

Image: Klinefelter Syndrome — Physical Characteristics in Affected vs. Unaffected Males
Cryptorchidism (Undescended Testes)
A history of undescended testicles may diminish fertility potential, even following surgical correction (orchiopexy). Extended time in the abnormal position can compound the impact on reproductive capacity. Medical guidelines recommend corrective surgery within the first year of life.[^11]
Infections
Infectious processes can disrupt sperm production, compromise sperm vitality, or create scar tissue that obstructs sperm passage:
Epididymitis and orchitis — Inflammatory conditions of the epididymis or testicular tissue that may injure structures responsible for sperm production.[^11]
Sexually transmitted infections — Untreated infections such as gonorrhea and chlamydia may trigger scarring and subsequent blockages.[^11]
Mumps orchitis — When mumps affects the testes in adult males, lasting damage and impaired fertility may follow.
Other Testicular Causes
Testicular cancer and treatment — Both the malignancy itself and therapeutic interventions (surgery, radiation, chemotherapy) can suppress sperm production.
Testicular trauma — Direct physical injury to testicular tissue may compromise sperm production.
Post-testicular Causes
Once produced, sperm must navigate through a series of ducts to exit the body. Post-testicular problems involve barriers along this pathway or difficulties with ejaculation.
→ Learn more: Azoospermia
Vas deferens obstruction — Blockages may result from prior vasectomy, infectious damage, or congenital absence of the tubes (CBAVD).[^4]
Ejaculatory duct obstruction — Cystic formations, prior infections, or structural irregularities can block these passages. This finding appears in approximately 5% of infertile males.
Retrograde ejaculation — Semen flows backward into the bladder rather than forward through the urethra. Contributing factors may include diabetes, spinal cord injury, certain medications, and prostate surgery.
Erectile dysfunction — Difficulty achieving or maintaining an erection sufficient for intercourse, which may indirectly impair fertility.

Image: Sperm Transport Pathway — Vas Deferens, Ejaculatory Duct, and Urethra
Pre-testicular Causes (Hormonal)
Pre-testicular factors disrupt the hormonal signals necessary for testicular function. The hypothalamic-pituitary-gonadal (HPG) axis orchestrates sperm production, and dysfunction at any level can impair this process.
→ Learn more: Male Hormonal Disorders
Hypogonadotropic hypogonadism — Insufficient production of FSH and LH — the hormones that drive testicular function. Pituitary tumors, Kallmann syndrome (often accompanied by anosmia), and other conditions can cause this deficiency. Treatable with hormone therapy.[^12]
Hyperprolactinemia — Elevated prolactin levels inhibit gonadotropin release, subsequently lowering testosterone and impairing sperm production.[^3]
Exogenous testosterone use — Taking testosterone from external sources — whether prescription therapy or anabolic steroids — suppresses the body’s own FSH and LH production, effectively halting sperm production. Effects may take months to years to reverse.
Obesity — Excess body weight increases the risk of hormonal disturbances and elevated scrotal temperature.
Thyroid disorders — Both hyperthyroidism and hypothyroidism may affect sperm development and hormonal balance.

Image: The HPG Axis — Hormonal Communication Between the Brain, Pituitary Gland, and Testes
What Environmental and Lifestyle Factors Affect Male Fertility?
External exposures and daily habits can exert considerable influence over sperm quantity and quality. Unlike inherited conditions, lifestyle factors are typically adjustable.
Risk Factor | Effect on Male Fertility |
Smoking | May damage sperm DNA, reduce sperm count, and motility |
Alcohol | May lower testosterone levels, cause erectile dysfunction, and decrease sperm production |
Drug use | Anabolic steroids, marijuana, cocaine, and opioids may reduce sperm quality and testosterone |
Heat exposure | Prolonged or frequent saunas, hot tubs, or laptop use on the lap may temporarily impair sperm production. Typically reversible once excessive heat exposure is discontinued |
Environmental toxins | Exposure to pesticides, heavy metals (lead, cadmium), and industrial chemicals has been associated with reduced sperm counts |
Obesity | May impact fertility through hormonal changes and increased scrotal temperature |
Source: Sharma R, et al. Cigarette smoking and semen quality. Eur Urol. 2016.[^8]
Key insight:
Tobacco use has been estimated to contribute to 13–15% of infertility cases overall.[^8] Research suggests that quitting smoking before fertility treatment is associated with improved outcomes through both natural conception and assisted reproduction.
What Are the Signs and Symptoms of Male Infertility?
For most affected men, unsuccessful attempts at conception represent the only indication of a fertility problem. Overt symptoms remain absent in the majority of cases. However, certain underlying disorders may manifest through observable signs:
Sexual function problems — Ejaculatory difficulties, diminished semen volume, reduced libido, or erectile challenges.
Testicular symptoms — Discomfort, swelling, or detectable lumps in the scrotal region.
Hormonal signs — Breast tissue enlargement (gynecomastia), sparse facial or body hair, or other markers of hormonal disruption.
Recurrent respiratory infections — May signal genetic syndromes such as cystic fibrosis or primary ciliary dyskinesia.
Inability to smell (anosmia) — A characteristic feature of Kallmann syndrome.
How Is Male Infertility Diagnosed?
A thorough fertility evaluation requires examination of both partners. Neglecting either person risks overlooking significant contributing factors. Notably, diagnostic procedures for men are generally simpler, faster, and less invasive than those for women.

Image: Sperm Cell Anatomy — Head, Midpiece, and Tail
Important:
Research indicates that approximately 6% of men undergoing infertility assessment are diagnosed with a significant underlying medical condition (such as hormonal disorders or, rarely, testicular cancer), underscoring the importance of thorough evaluation.[^12]
Semen Analysis (Spermiogram)
The semen analysis forms the foundation of male fertility assessment. Given substantial natural variation in semen characteristics, guidelines recommend collecting at least 2 samples, spaced 2–4 weeks apart; if the second sample is abnormal, or the patient has suffered a fever or illness, repeat after approximately 3 months to complete the spermatogenesis cycle.[^5]
→ Diagnostic test: Semen Analysis (Spermiogram)

Image: Semen Analysis — Sample Collection Procedure
WHO 2021 Reference Values
Parameter | Lower Reference Limit |
Semen volume | ≥1.4 mL |
Sperm concentration | ≥16 million/mL |
Total sperm count | ≥39 million per ejaculate |
Total motility | ≥42% |
Progressive motility | ≥30% |
Vitality (live sperm) | ≥54% |
Normal morphology | ≥4% |
Source: WHO Laboratory Manual for the Examination and Processing of Human Semen, 6th edition (2021).[^5]
Semen Analysis Terminology
Term | Definition |
Azoospermia | No sperm in the ejaculate |
Oligozoospermia | Sperm concentration <16 million/mL |
Asthenozoospermia | Progressive motility <30% |
Teratozoospermia | Abnormal morphology <4% |
OAT syndrome | A combination of low count, motility, and morphology abnormalities |
Necrozoospermia | All sperm are non-viable |
Source: WHO Laboratory Manual for the Examination and Processing of Human Semen, 6th edition (2021).[^5]

Image: Sperm Count — Normal Concentration (Left) vs. Oligozoospermia (Right)

Image: Sperm Morphology — Normal Sperm (Left) vs. Midpiece Defects, Tail Defects (Top Row), Head Defects (Bottom Row)
Hormonal Testing
A blood hormone assessment is warranted when semen results show abnormalities or when clinical examination suggests an endocrine issue.
→ Diagnostic test: Hormonal Panel for Infertility
Hormone | Function | Clinical Significance |
FSH | Stimulates Sertoli cells and spermatogenesis | Elevated = testicular damage; Low = pituitary problem |
LH | Stimulates Leydig cells to produce testosterone | Helps differentiate primary vs. secondary hypogonadism |
Testosterone | Essential for spermatogenesis | Low levels indicate hypogonadism |
Prolactin | Elevated levels suppress gonadotropins | High = possible pituitary tumor |
Estradiol | Regulates feedback to the pituitary | Elevated = may benefit from an aromatase inhibitor |
Source: ASRM Practice Committee. Diagnostic evaluation of the infertile male. Fertil Steril. 2006.[^4]
Genetic Testing
Chromosomal and genetic screening is advisable when sperm concentration falls below 5 million/mL or when no sperm appear in the ejaculate. Chromosomal abnormalities occur far more frequently among infertile men with azoospermia or severe oligozoospermia (up to 15%) compared with fertile counterparts (approximately 0.6%).[^10]
Karyotype analysis—Detects chromosomal abnormalities, including Klinefelter syndrome.
Y-chromosome microdeletion testing—Indicated when sperm counts fall below 5 million/mL.
CFTR gene testing—Appropriate when physical examination reveals absent vas deferens.
Imaging Studies
Scrotal ultrasound examination can reveal varicoceles, testicular masses, epididymal abnormalities, and enable volumetric measurement of the testes. For azoospermic patients with reduced semen volume and absent or abnormal vas deferens, transrectal ultrasound (TRUS) may help assess potential ejaculatory duct obstruction.
What Are the Treatment Options for Male Infertility?
Treatment strategy depends on the identified cause, degree of semen abnormality, and the couple’s preferences. The standard approach follows a graduated pathway, beginning with conservative measures and advancing to more intensive interventions as needed.
Lifestyle Modifications: The First Step
Regardless of diagnosis, optimizing overall health may enhance reproductive potential:
Eliminate smoking and vaping.
Reduce or eliminate alcohol consumption.
Avoid cannabis, anabolic steroids, and illicit substances.
Achieve and maintain optimal body weight through balanced nutrition and physical activity.
Avoid prolonged exposure to heat, such as hot baths and saunas.
Limit contact with pesticides, industrial pollutants, and heavy metals.
Medical Treatment
Hormonal Therapy
Pharmaceutical hormone interventions may prove particularly effective for select diagnoses:
Gonadotropin therapy (FSH, hCG) — May deliver excellent results in hypogonadotropic hypogonadism, with studies suggesting that up to 80% of treated men may experience restoration of spermatogenesis.[^12]
Clomiphene citrate — This antiestrogen compound elevates gonadotropin levels and may promote sperm development in appropriate candidates.
Aromatase inhibitors (anastrozole, letrozole) — May be beneficial for men with elevated estradiol levels and/or low testosterone to estradiol ratio.[^7]
Dopamine agonists — Agents such as cabergoline can normalize prolactin levels in patients with hyperprolactinemia, thereby restoring reproductive function.[^3]
Recovery from Testosterone Therapy
Most men recover sperm production within 12–24 months after discontinuation, although recovery time varies and may be incomplete in some cases.[^12] This requires stopping testosterone and working with an endocrinologist or reproductive urologist.
Important:
Supplemental testosterone and anabolic steroids rank among the most common causes of self-induced male infertility. The encouraging news: recovery typically follows discontinuation.
Surgical Treatment
→ Learn more: Surgical Solutions for Infertility
Varicocele Correction (Varicocelectomy)
Varicocelectomy is generally considered for infertile men with both detectable varicoceles on examination and suboptimal semen parameters. Research suggests surgical intervention may yield improved semen quality in 60–70% of men with clinically significant varicoceles.[^6]
Vasectomy Reversal
Men who previously underwent vasectomy may pursue fertility restoration through microsurgical reconnection (vasovasostomy or vasoepididymostomy). Outcomes correlate strongly with elapsed time — those whose vasectomy occurred within the preceding decade may achieve sperm patency rates approaching 90%.[^13]
Sperm Retrieval Procedures
When obstruction or primary testicular failure results in absent ejaculated sperm, surgical extraction directly from reproductive tissues may succeed:
TESA (Testicular Sperm Aspiration)—A needle-based technique extracting tissue from the testis.
Micro-TESE (Microsurgical Testicular Sperm Extraction)—Considered optimal for non-obstructive azoospermia; employs operative microscopy to locate pockets of active sperm production.
PESA (Percutaneous Epididymal Sperm Aspiration)—Suited for cases of obstructive azoospermia.
Assisted Reproductive Technology (ART)
When conventional treatments prove insufficient—or are not appropriate—assisted reproductive methods offer highly effective alternatives.
Method | What It Is | Success Rate (Clinical Pregnancy Rate per Cycle) | Best For |
IUI | Prepared sperm is placed directly into the uterus using a thin catheter at the time of ovulation | 4–17% per cycle (clinical pregnancy rate; 4% alone, up to 17% with ovarian stimulation) | Mild male factor, unexplained infertility |
IVF | Oocytes are retrieved and fertilized in a laboratory setting, where sperm are added under controlled conditions, and embryo(s) are transferred to the uterus | 40–50% per cycle (<35 years, clinical pregnancy rate); declines with age | Moderate male factor, blocked tubes |
ICSI | A single sperm is injected directly into each oocyte | Similar to IVF; fertilization rate 50–80% | Severe male factor, azoospermia with sperm retrieval |
Source: CDC. 2021 ART Fertility Clinic and National Summary Report. 2023; ASRM Practice Committee Guidelines.[^9]
ICSI fundamentally transformed outcomes for severe male factor infertility. Today, even men with only a handful of functional sperm — extracted surgically from testicular tissue — can become biological fathers. Studies report ICSI achieves fertilization in approximately 50–80% of injected mature oocytes, depending on sperm quality and other factors.[^9]
→ Learn more: Assisted Reproductive Technology
What Is the Prognosis for Male Infertility?
Expected outcomes in male infertility span a broad range based on the specific underlying disorder:
Category | Proportion of Cases |
Treatable causes (obstruction, hormonal deficiency, varicocele) | Approximately 18% |
Subfertility requiring ART (oligozoospermia, asthenozoospermia) | Approximately 70% |
Untreatable sterility (primary testicular failure) | Approximately 12% |
Source: Barak S, Baker HWG. Clinical Management of Male Infertility. Endotext. 2016.[^17]
Encouraging news:
Nature sometimes finds a way — research suggests approximately 23% of couples diagnosed with infertility may conceive within 2 years without medical intervention, rising to 33% over 4 years. Remarkably, even men with severely reduced counts below 2 million/mL may carry a 7.6% probability of achieving pregnancy within 2 years naturally.[^14]
How Can Male Infertility Be Prevented?
While specific causes lie beyond anyone’s control, men can actively reduce several established risk factors:
Refrain from smoking tobacco products and vaping.
Keep alcohol consumption moderate or eliminate it.
Avoid recreational drugs and performance-enhancing steroids.
Maintain a healthy body weight.
Avoid prolonged exposure of the testicles to high temperatures.
Minimize exposure to pesticides, industrial chemicals, and heavy metals.
Use barrier protection during intercourse to guard against fertility-affecting sexually transmitted infections (STIs).
Consider fertility preservation options before undergoing cancer therapy.
Is Male Infertility a Marker of Overall Health?
Research indicates that men experiencing fertility difficulties show elevated rates of various cancers (particularly testicular malignancies), autoimmune conditions, and cardiovascular disease relative to their fertile peers.[^14][^15]
This relationship elevates the importance of fertility evaluation beyond reproduction alone — it may serve as a window into general health status.[^12]
So, What Should You Do Now?
For couples facing conception challenges, here’s a practical roadmap:
Step 1: Determine If It’s Time to Seek Help
Trying for over a year? Time for both partners to undergo assessment.
Female partner 35 or older? Don’t wait longer than 6 months.
Pre-existing risk factors present? (varicocele, history of undescended testicles, past infections) Pursue evaluation promptly.
Step 2: Get Evaluated—Both Partners
For him: Semen Analysis (Spermiogram)—straightforward, affordable, and capable of detecting the most significant sperm abnormalities.
For her: Hormonal panel, ultrasound, and potentially hysterosalpingography (HSG).
Step 3: Understand Your Diagnosis
With a precise diagnosis, you gain the ability to make informed decisions among treatment options. Effective solutions exist for the vast majority of identified causes.
Step 4: Explore Your Treatment Options
Your treatment path depends on what testing reveals: lifestyle modifications, medication, surgery, IUI, IVF, or ICSI.
Step 5: Choose the Right Clinic
When assisted reproduction becomes necessary, clinic selection matters greatly. Consider factors such as published success statistics, the breadth of services offered, pricing structure, and practitioner qualifications.
→ Compare fertility clinics worldwide: MedicalNavigator.com/fertility-clinics
Too Long, Didn’t Read
Male factors are involved in 40–50% of all infertility cases.
Semen analysis is quick, non-invasive, and reveals the most common problems.
Varicocele is the most common treatable cause, found in up to 40% of infertile men.
Lifestyle changes can improve outcomes: stop smoking, moderate alcohol, and maintain a healthy weight.
Most causes are treatable or can be bypassed with assisted reproduction.
First step: get a semen analysis—it’s simple and answers many questions.
References
[^1]: World Health Organization. Infertility Fact Sheet. November 2023.
[^2]: Agarwal A, Mulgund A, Hamada A, Chyatte MR. A unique view on male infertility around the globe. Reprod Biol Endocrinol. 2015;13:37.
[^3]: Melmed S, Casanueva FF, Hoffman AR, et al. Diagnosis and treatment of hyperprolactinemia: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-288.
[^4]: Practice Committee of the ASRM. Report on optimal evaluation of the infertile male. Fertil Steril. 2006;86(5 Suppl 1):S202-S209.
[^5]: World Health Organization. WHO laboratory manual for the examination and processing of human semen, 6th edition. 2021.
[^6]: Jensen CFS, Sønksen J, Fode M, et al. Varicocele and male infertility. Nat Rev Urol. 2017;14(9):523-533.
[^7]: Schlegel PN. Aromatase inhibitors for male infertility. Fertil Steril. 2012;98(6):1359-1362.
[^8]: Sharma R, Harlev A, Agarwal A, Esteves SC. Cigarette smoking and semen quality: a new meta-analysis examining the effect of the 2010 WHO laboratory manual. Eur Urol. 2016;70(4):635-645.
[^9]: Centers for Disease Control and Prevention. 2021 ART Fertility Clinic and National Summary Report. 2023.
[^10]: Zorrilla M, Yatsenko AN. The genetics of infertility: current status of the field. Curr Genet Med Rep. 2013;1(4):247-260.
[^11]: Bachir BG, Jarvi K. Infectious, inflammatory, and immunologic conditions resulting in male infertility. Urol Clin North Am. 2014;41(1):67-81.
[^12]: Leslie SW, Soon-Sutton TL, Khan MA. Male Infertility. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024.
[^13]: Belker AM, Thomas AJ, Fuchs EF, Konnak JW, Sharlip ID. Results of 1,469 microsurgical vasectomy reversals by the Vasovasostomy Study Group. J Urol. 1991;145(3):505-511.
[^14]: Del Giudice F, Kasman AM, Ferro M, et al. Clinical correlation among male infertility and overall male health: a systematic review of the literature. Investig Clin Urol. 2020;61(4):355-371.
[^15]: Salonia A, Matloob R, Gallina A, et al. Are infertile men less healthy than fertile men? Results of a prospective case-control survey. Eur Urol. 2009;56(6):1025-1031.
[^16]: Winters BR, Walsh TJ. The epidemiology of male infertility. Urol Clin North Am. 2014;41(1):195-204.
[^17]: Barak S, Baker HWG. Clinical Management of Male Infertility. In: Feingold KR, et al., editors. Endotext [Internet]. MDText.com, Inc.; South Dartmouth (MA): Feb 5, 2016.
This guide is for informational purposes only. Always consult qualified healthcare providers for personalized recommendations. For full details, read our Medical Disclaimer.
Author of the article
Date of publication









